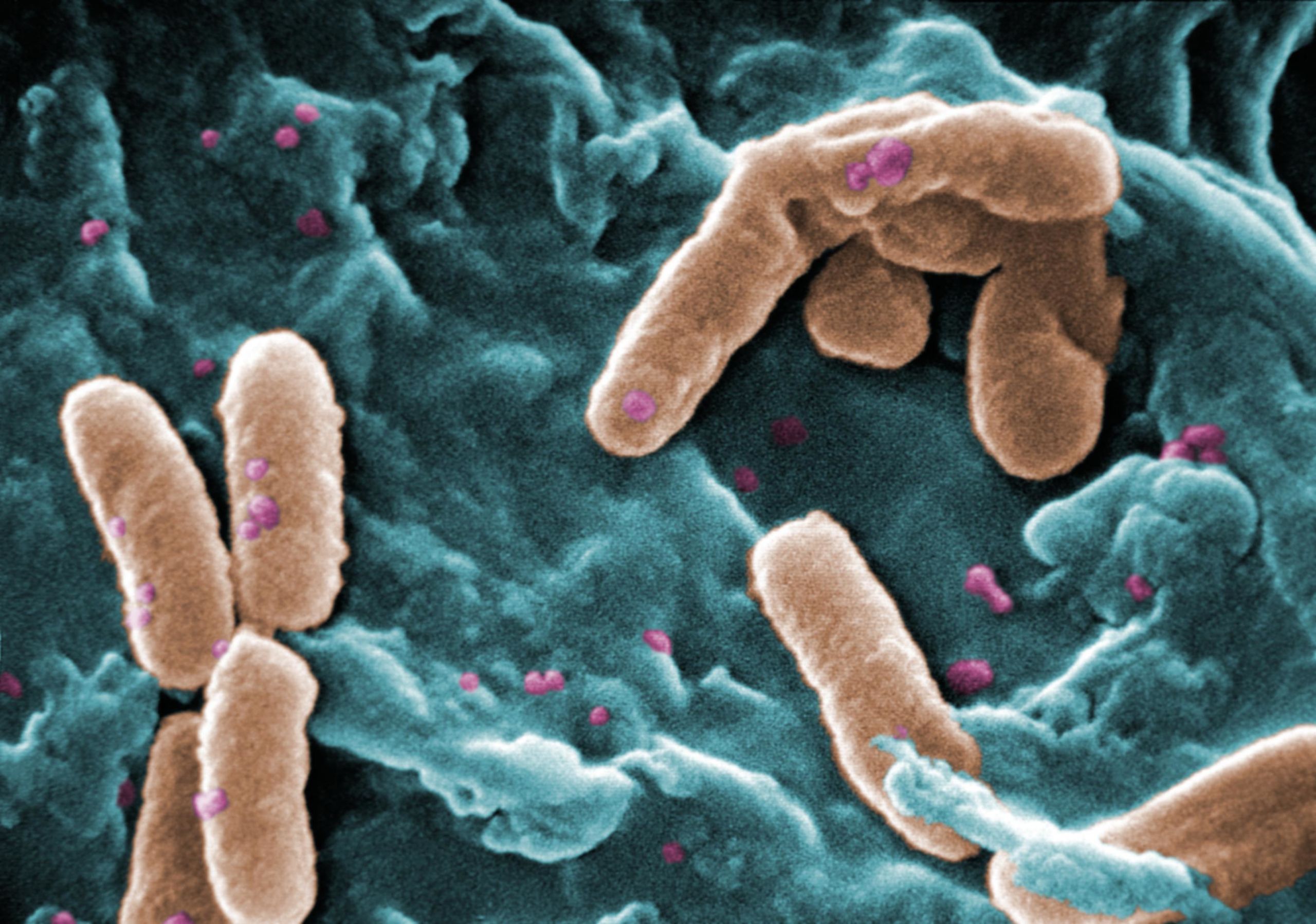
Pseudomonas aeruginosa is one of the main causes of pneumonia in immunosuppressed patients and people with lung diseases. Due to its high level of antibiotic resistance, it is increasingly difficult to eradicate the infections it causes and consequently, the WHO (World Health Organization) considers the development of new antibiotics against this pathogen a priority.
One of the factors that make Pseudomonas aeruginosa lethal is the Type VI Secretion System (T6SS). This secretory system assembles within Pseudomonas aeruginosa, and, when in contact with other cells, uses a contractile molecular mechanism to inject toxins into them. Although in the last decade a great advance has been made in our knowledge about Pseudomonas aeruginosa T6SS, and its relevance in bacterial competition and pathogenesis, the identities and functions of the vast majority of toxins secreted by T6SS are still unknown.
An international research team from the Institute of Biofisika (CSIC-UPV / EHU) led by David Albesa, has identified for the first time several toxins secreted by T6SS, among which is Tse8. This discovery may have effects on the future development of antibiotics. In the words of David Albesa, “The development of new antibiotics can be significantly facilitated if the biology of the causative agents is better understood. In this sense, Tse8 causes cell death in other bacteria in its environment, so deciphering the mechanism of action at the molecular level of this toxin could help to develop innovative strategies to combat pathogenic bacteria ”.
The work has been published in the prestigious Journal of Structural Biology.
Link to the scientific publication: https://doi.org/10.1016/j.jsb.2020.107651.
.png)
